Indication Aimovig® is indicated for the preventive treatment of migraine in adults.
Indication Aimovig® is indicated for the preventive treatment of migraine in adults.
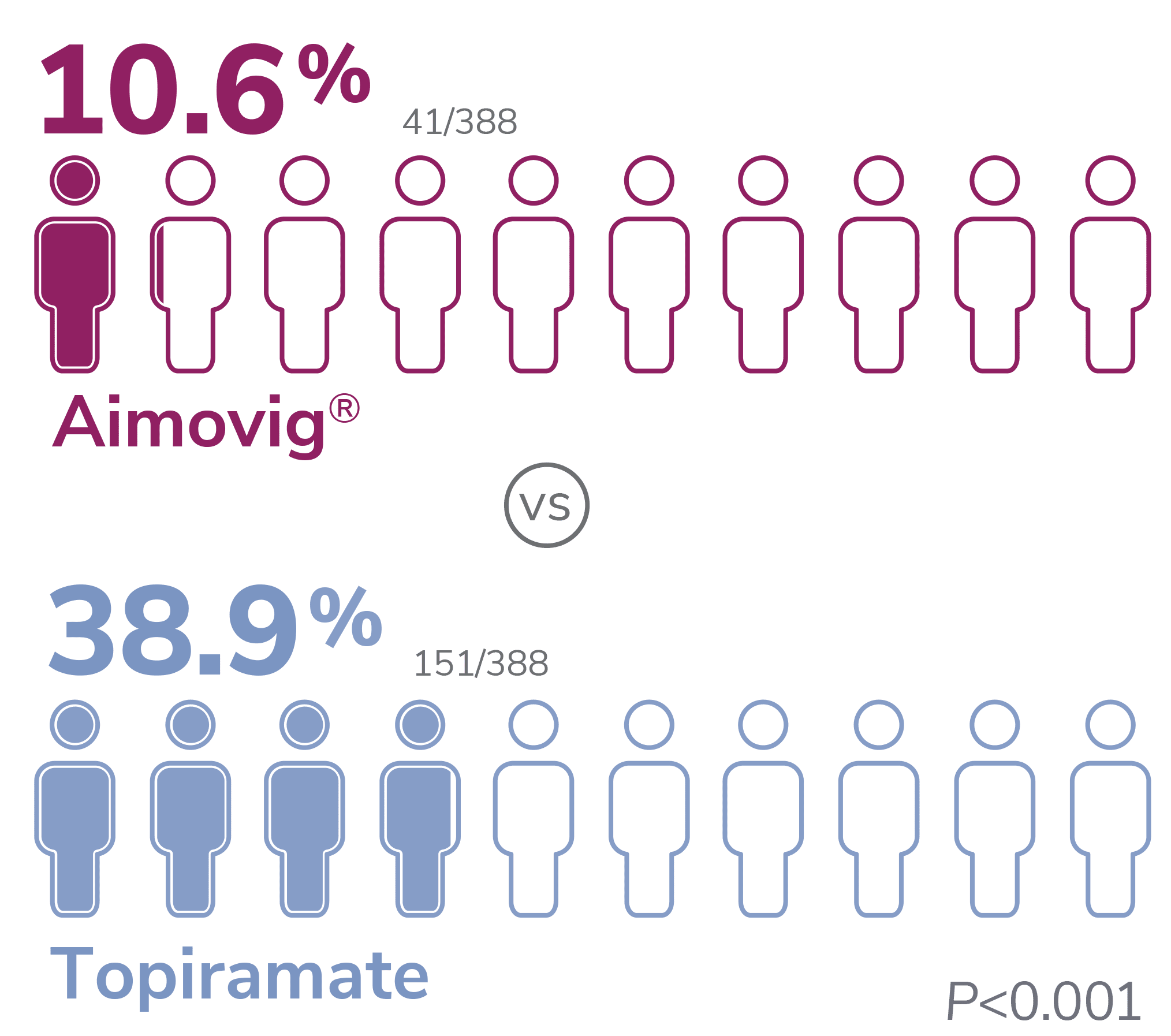
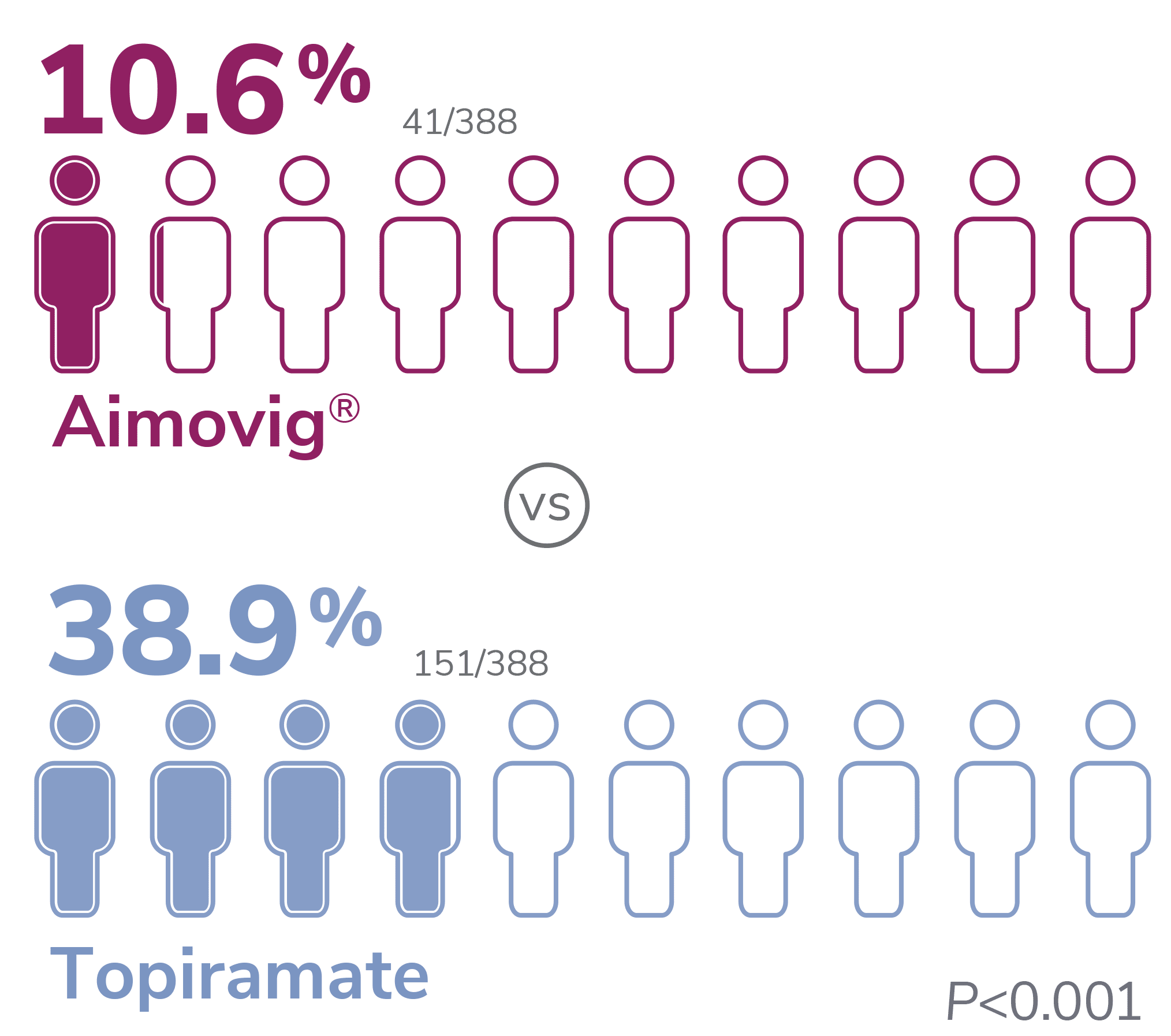
With Aimovig®, the most frequent AEs leading to discontinuation of study medication were fatigue (2.3%), nausea (2.1%), disturbance in attention (1.8%), and dizziness (1.0%). In the topiramate group, they were parasthesia (9.8%), disturbance in attention (9.3%), fatigue (7.5%), and nausea (6.7%)1
HER-MES was a double-blind, double-dummy, randomized, multicenter, 24-week phase 4 study of adult patients with episodic or chronic migraine. Patients were randomized 1:1 to receive Aimovig® 70 mg or 140 mg, based on investigator decision, plus topiramate placebo or oral topiramate (50/75/100 mg) at the individual dose with optimal efficacy plus Aimovig® placebo following a 6-week titration phase. If response was insufficient for patients receiving Aimovig® 70 mg, the dose could be increased to 140 mg at any point during the 6-month DBTP. Dose reduction was not permitted for either drug.1

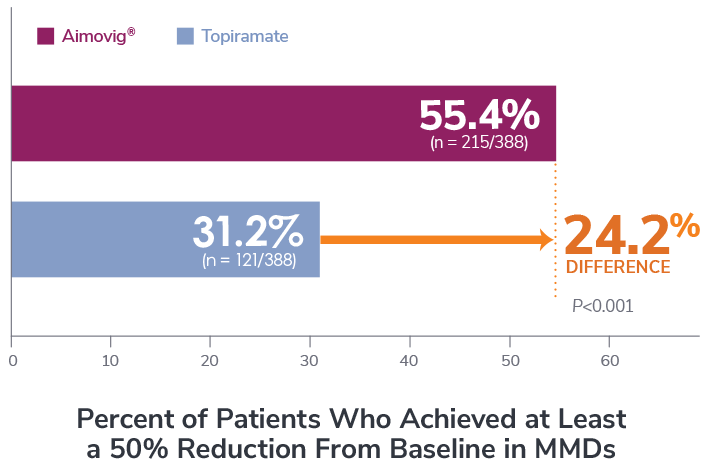
P-value was obtained based on odds of achieving ≥50% reduction in MMDs which were significantly higher for the Aimovig® group than the topiramate group (Odds Ratio = 2.76; 95% CI: 2.06, 3.71).1
*Patients who discontinued study treatment could remain in the study for further data collections. Patients with missing eDiary entries in all Months 4-6 visits were imputed as non-response.1
P-value was obtained based on odds of achieving ≥50% reduction in MMDs which were significantly higher for the Aimovig® group than the topiramate group (Odds Ratio = 2.76; 95% CI: 2.06, 3.71).1
*Patients who discontinued study treatment could remain in the study for further data collections. Patients with missing eDiary entries in all months 4-6 visits were imputed as non-response.1
Aimovig® (erenumab-aooe) is indicated for the preventive treatment of migraine in adults.
Contraindication: Aimovig® is contraindicated in patients with serious hypersensitivity to erenumab-aooe or to any of the excipients. Reactions have included anaphylaxis and angioedema.
Hypersensitivity Reactions: Hypersensitivity reactions, including rash, angioedema, and anaphylaxis, have been reported with Aimovig® in post marketing experience. Most reactions were not serious and occurred within hours of administration, although some occurred more than one week after administration. If a serious or severe reaction occurs, discontinue Aimovig® and initiate appropriate therapy.
Constipation with Serious Complications: Constipation with serious complications has been reported following the use of Aimovig® in the postmarketing setting. There were cases that required hospitalization, including cases where surgery was necessary. The onset of constipation was reported after the first dose in a majority of these cases, but patients also reported later on in treatment. Aimovig® was discontinued in most reported cases. Constipation was one of the most common (up to 3%) adverse reactions reported in clinical studies.
Monitor patients treated with Aimovig® for severe constipation and manage as clinically appropriate. Concurrent use of medications associated with decreased gastrointestinal motility may increase the risk for more severe constipation and the potential for constipation-related complications.
Hypertension: Development of hypertension and worsening of pre-existing hypertension have been reported following the use of Aimovig® in the postmarketing setting. Many of the patients had pre-existing hypertension or risk factors for hypertension. There were cases requiring pharmacological treatment and, in some cases, hospitalization. Hypertension may occur at any time during treatment but was most frequently reported within seven days of dose administration. In the majority of the cases, the onset or worsening of hypertension was reported after the first dose. Aimovig® was discontinued in many of the reported cases.
Monitor patients treated with Aimovig® for new-onset hypertension, or worsening of pre-existing hypertension, and consider whether discontinuation of Aimovig® is warranted if evaluation fails to establish an alternative etiology.
Adverse Reactions: The most common adverse reactions in clinical studies (≥ 3% of Aimovig®-treated patients and more often than placebo) were injection site reactions and constipation.
Please see Aimovig® full Prescribing Information.
Contraindication: Aimovig® is contraindicated in patients with serious hypersensitivity to erenumab-aooe or to any of the excipients. Reactions have included anaphylaxis and angioedema.
Hypersensitivity reactions: Hypersensitivity reactions, including rash, angioedema, and anaphylaxis, have been reported with Aimovig® in post marketing experience. Most reactions were not serious and occurred within hours of administration, although some occurred more than one week after administration. If a serious or severe
References: 1. Reuter U, Ehrlich M, Gendolla A, et al. Erenumab versus topiramate for the prevention of migraine–a randomised, double-blind, active-controlled phase 4 trial. Cephalalgia. 2021. doi:10.1177/03331024211053571. 2. Data on file, Amgen; 2024.